There have been many studies discussing the mineral alterations in CO 2-saturated NaCl saline systems at different pressures and temperatures ( Ueda et al., 2021 Edem et al., 2022 Peter et al., 2022), however, many questions regarding reaction mechanisms are yet to be answered.  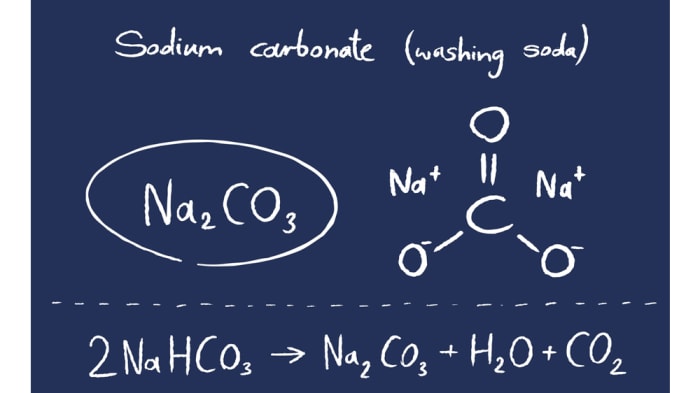
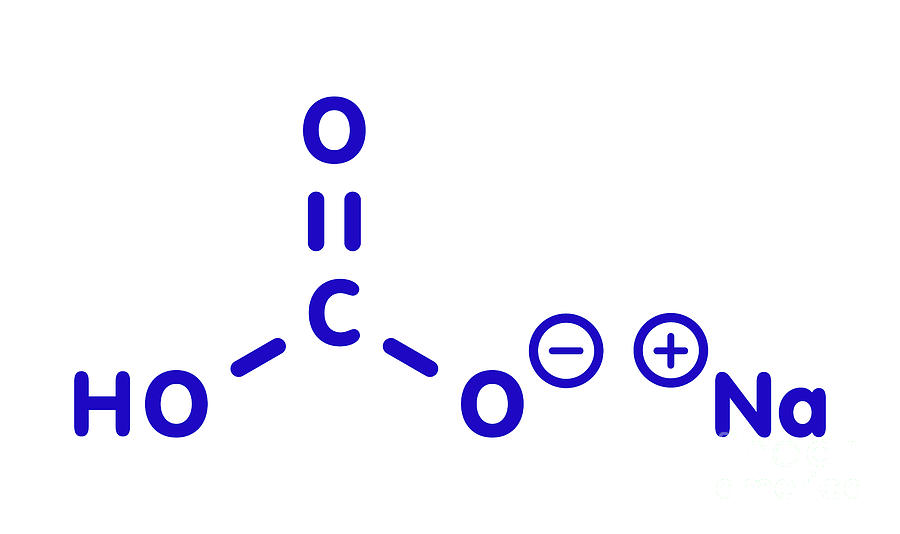
To alleviate water shortage, recently, researchers suggested substituting freshwater with saline water (e.g., seawater) for long-term CO 2 storage ( Voigt et al., 2021), which makes the understanding of CO 2-saline water-basalt interactions even more important. However, it has been estimated that 22 tons of water is required to capture 1 ton of pure CO 2 for storage according to the operation in CarbFix ( Snæbjörnsdóttir et al., 2020). This successful application is inspiring other CO 2 mineralization projects around the world to be developed. Basalt is one of the optimum rocks for CO 2 storage because of its chemical composition, abundant distribution and easy availability.ĬarbFix Project in Iceland is an engineering example for large-scale CO 2 mineralization, where 95% of the dissolved CO 2 in freshwater is mineralized to carbonates in the basaltic groundwater setting which has a weakly alkaline pH and temperature of 20☌–50☌ in 2 years ( Alfredsson et al., 2013 Matter et al., 2016 Snæbjörnsdóttir et al., 2020). Among the CCUS techniques, mineralization of CO 2 to water-insoluble carbonates using Mg or Ca-rich rocks such as basalt ( Goldberg et al., 2008 Okoko and Olaka, 2021), peridotite ( Oelkers et al., 2008) and wollastonite ( Wang et al., 2021) is a promising method for large-scale and long-term CO 2 storage with low risk of releasing stored CO 2 into the atmosphere ( Zhao et al., 2015) and probably a relative acceptable cost ( National Academies of Sciences, 2018 Kelemen et al., 2019). The urgent need to reduce the amount of carbon dioxide (CO 2) for creating a carbon neutral society has driven the development of various CO 2 capture, utilization, and storage (CCUS) techniques ( Voigt et al., 2021). This study illuminated the role of NaHCO 3 in basalt carbonation and provided technical backup to the design of advanced CO 2 mineralization systems. Due to the presence of additional Na in solution, all the released elements were consumed quickly with generation of secondary minerals in turn promoted basalt dissolution to release more Ca for CO 2 storage. In such hydrothermal systems, basalt was carbonated to generate calcite (CaCO 3), where the Ca was mainly from plagioclase Mg and Fe were incorporated into smectite, and Na in the saline system participated in the formation of Na silicates (i.e., analcime in the case of basalt). 
As revealed by experiments conducted at the saturated vapor pressure of water, the carbon storage efficiency at 300☌ reached 75 g/kg of basalt in 5 days, which was 12 times higher than that at 200☌. The inclusion of NaHCO 3 was confirmed to drastically promote the alteration of basalt, especially at higher temperatures. Considering multiple potential positive effects of the CO 2 carrier, NaHCO 3, we conducted this study to experimentally evaluate the CO 2 storage efficiency during water-basalt-NaHCO 3 interactions under hydrothermal conditions at 200–300☌. As a promising feedstock, basaltic rock has gained special interest, and elevating basalt carbonation efficiency with the reduction of negative environmental impact is the main challenge for CO 2 mineralization system development. Reaction of carbon dioxide (CO 2) with minerals to generate stable carbonates, also known as CO 2 mineralization, has been regarded as one of the most promising methods for safe and permanent carbon storage.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
